Inhibitory receptors play an important role in controlling the immune response. Physiologically, the role of inhibitory receptors is to decrease the immune response thereby resolving inflammation. Because of this function, inhibitory receptors are often referred to as immune checkpoints. We develop monoclonal antibodies that activate or block inhibitory receptors on immune cells. Activating inhibitory receptors with agonist antibodies allows us to selectively inhibit cells involved in disease pathogenesis and, in the setting of allergy and inflammation, has the potential to reduce inflammation. In addition, agonist antibodies that target inhibitory receptors have the distinct advantage of counteracting multiple activation pathways and therefore have the potential to reduce inflammation more broadly than targeting a single cytokine or receptor pathway. Moreover, because tumors can utilize inhibitory receptors to avoid detection by the immune systems, blocking an inhibitory receptor with an antagonist antibody can restore the immune system’s ability to identify and kill tumor cells.
Our Lead Anti-Inflammatory Program Targets Mast Cells
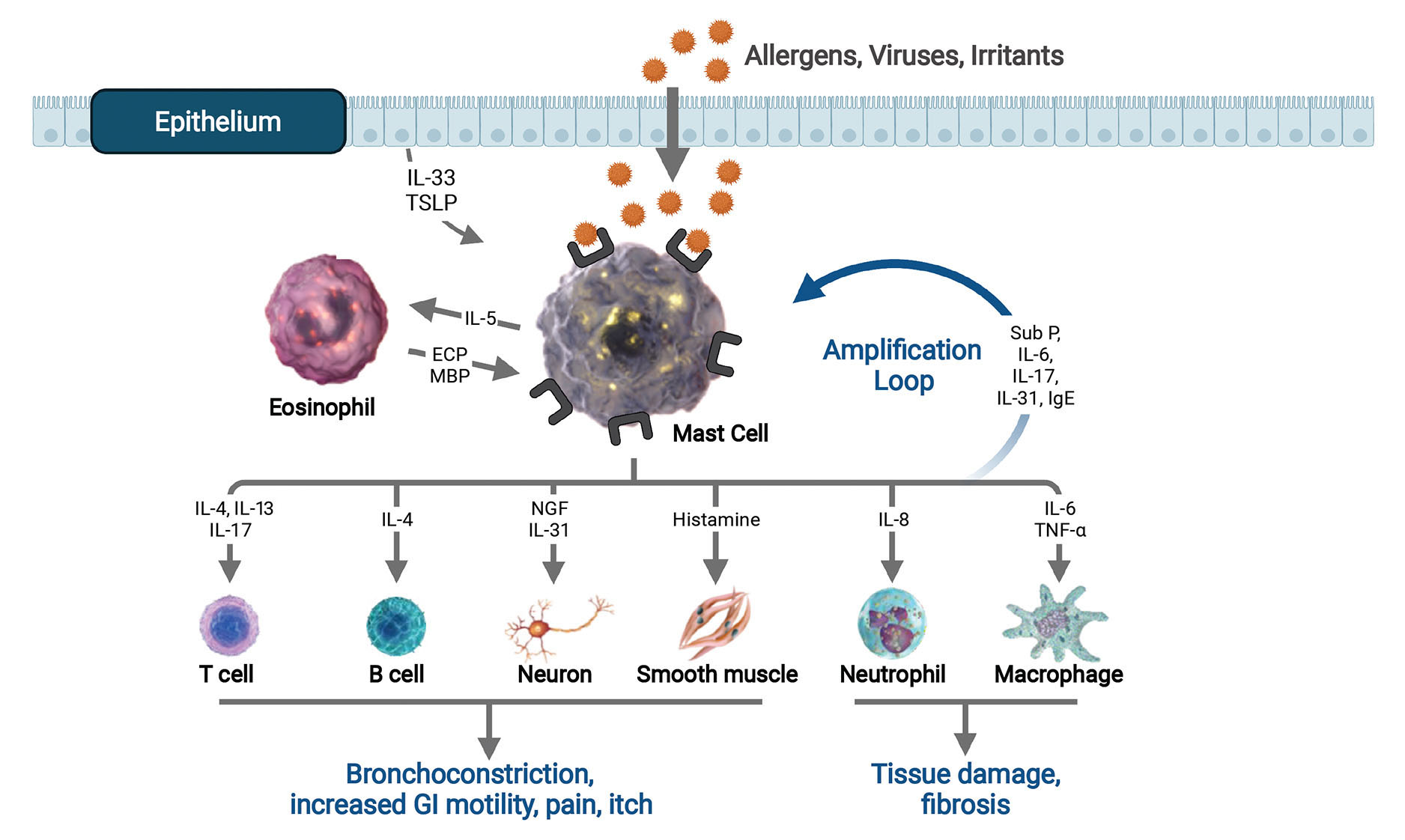
Mast cells are found at the internal/external interfaces of the body, in particular at mucosal surfaces, surrounding blood vessels, and in close proximity to peripheral nerves. They produce a broad range of inflammatory mediators including vasoactive amines, bioactive lipids, proteases, cytokines and chemokines and express many immunologically important cell surface receptors. As a result, mast cells are involved in both innate and adaptive immune responses, and participate in both acute and chronic inflammation. Because of the inflammatory mediators they release and their ability to recruit and activate other immune cells through expression of chemokines and cytokines, mast cells are believed to drive symptoms in a number of diseases. Agents targeting mast cell receptors or secretion products have shown utility in the treatment of a number of diseases including chronic urticaria, atopic dermatitis, severe asthma, chronic rhinosinusitis, eosinophilic gastrointestinal diseases, indolent systemic mastocytosis, highlighting pathogenic roles of these cells. By targeting inhibitory receptors on mast cells, Allakos is seeking to produce drugs with advantages over agents that target a single secretion product or receptor pathway.
Mast Cells are Key Drivers of Inflammatory Disease
AK006 (Siglec-6 directed agonist monoclonal antibody)
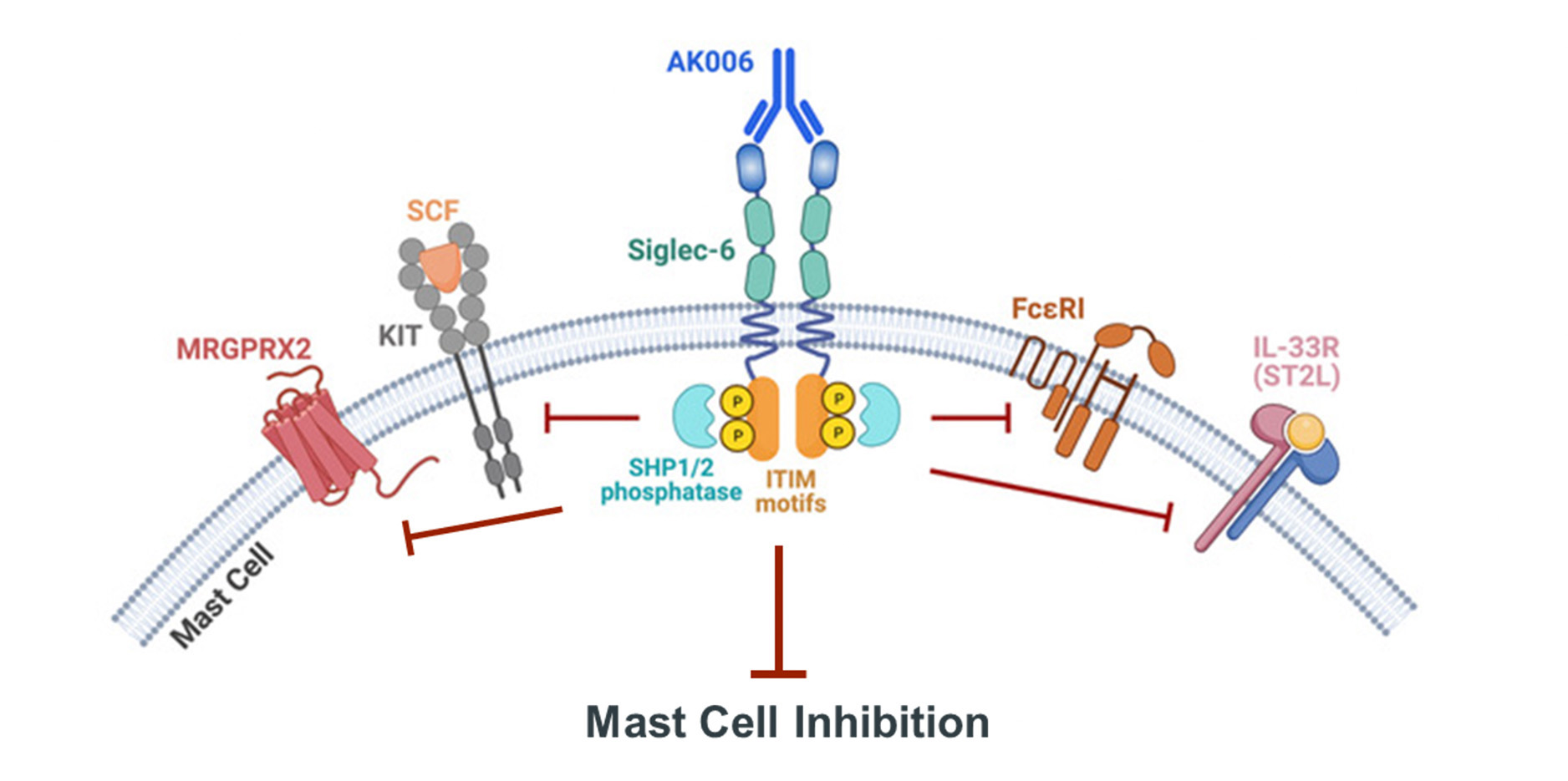
Siglec-6 is a member of the family of cell surface receptors called Sialic acid-binding immunoglobulin-type lectins (Siglecs). Siglec-6 is found on the surface of mature mast cells, and therefore offers a way to target mast cells. Siglec-6 exerts inhibition through its intracellular immunoreceptor tyrosine-based motif (ITIM).
ITIM bearing receptors antagonize activating receptors and consequently have important roles in regulating the immune system. The inhibitory function is derived from the ability of the ITIMs to recruit SH2 domain-containing phosphatases which work to oppose activating signals driven by kinase signaling cascades. Disrupting kinase signaling cascades has been a successful strategy for treating inflammatory diseases as evidenced by approved drugs which target JAK, KIT, BTK, SYK, and others. However, often these kinase signaling pathways are active in multiple cell types, which can result in unintended side effects when disrupted.
AK006 is a humanized IgG1 monoclonal antibody which activates the inhibitory receptor Siglec-6. AK006 is directed to an extracellular epitope of the Siglec-6 receptor that was identified for its ability to generate strong inhibitory signals to mast cells. Furthermore, AK006 was engineered to have higher cell surface residence time which may increase mast cell inhibition. In addition to inhibition, in preclinical studies AK006 reduces mast cell numbers via antibody-dependent cellular phagocytosis (ADCP) in the presence of activated macrophages.
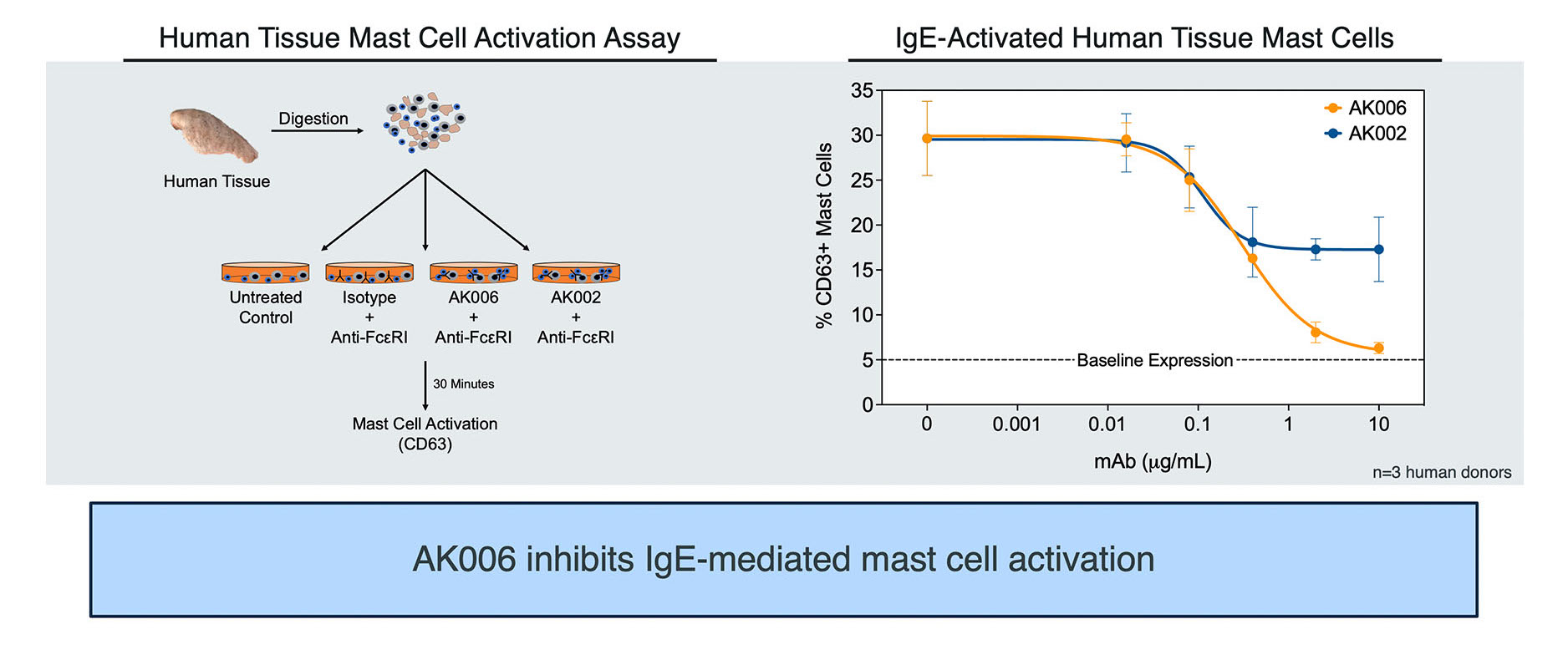
AK006 shows deep mast cell inhibition
In vitro and in vivo studies have demonstrated AK006 inhibits multiple modes of mast cell activation including IgE, IL-33, KIT, C5a, and MRGPRX2, resulting in the broad suppression of inflammation. In preclinical studies, AK006 displays significantly stronger mast cell inhibition than AK002.
AK006 Inhibits Mast Cell Activation in Human Tissues
AK006 is currently being studied in Healthy Volunteers and Allakos plans to initiate a Phase 1 trial in patients with Chronic Spontaneous Urticaria (CSU) in Q2 2024
AK007 (Siglec-10 directed antagonist monoclonal antibody)
In proliferative diseases like cancer, blocking immune inhibitory checkpoint receptors can restore the immune system’s ability to identify and kill tumor cells. Therapeutics that target T cell checkpoint receptors, such as PD-1 and CTLA-4, or their ligands, were the first to demonstrate meaningful anti-tumor activity by blocking immune cell inhibition (i.e. removing the brakes on the immune system). More recently, ‘don’t eat me’ signals, such as CD47 and CD24, have been identified which allow cancer cells to avoid destruction by macrophages and other myeloid cells of the innate immune system. Restoring myeloid cell function has the potential to increase anti-tumor immunity by activating both innate and adaptive immune cells.
Siglec-10 is a checkpoint receptor selectively expressed on tumor associated macrophages (TAMs) and dendritic cells (DCs). It functions as an inhibitory receptor through interaction with multiple ligands, including the ‘don’t eat me’ signal CD24 as well as CD52 and VAP-1. Siglec-10 activation induces immunosuppression and promotes tumor immune escape through interaction with CD24. Similarly, CD52 has been shown to induce inhibition via Siglec-10, indicating that Siglec-10 functions as an inhibitory receptor through multiple ligands. Siglec-10 is elevated in multiple tumor types and increased expression has been inversely correlated with patient survival in multiple solid tumors, suggesting that Siglec-10 may play a role in tumor evasion.
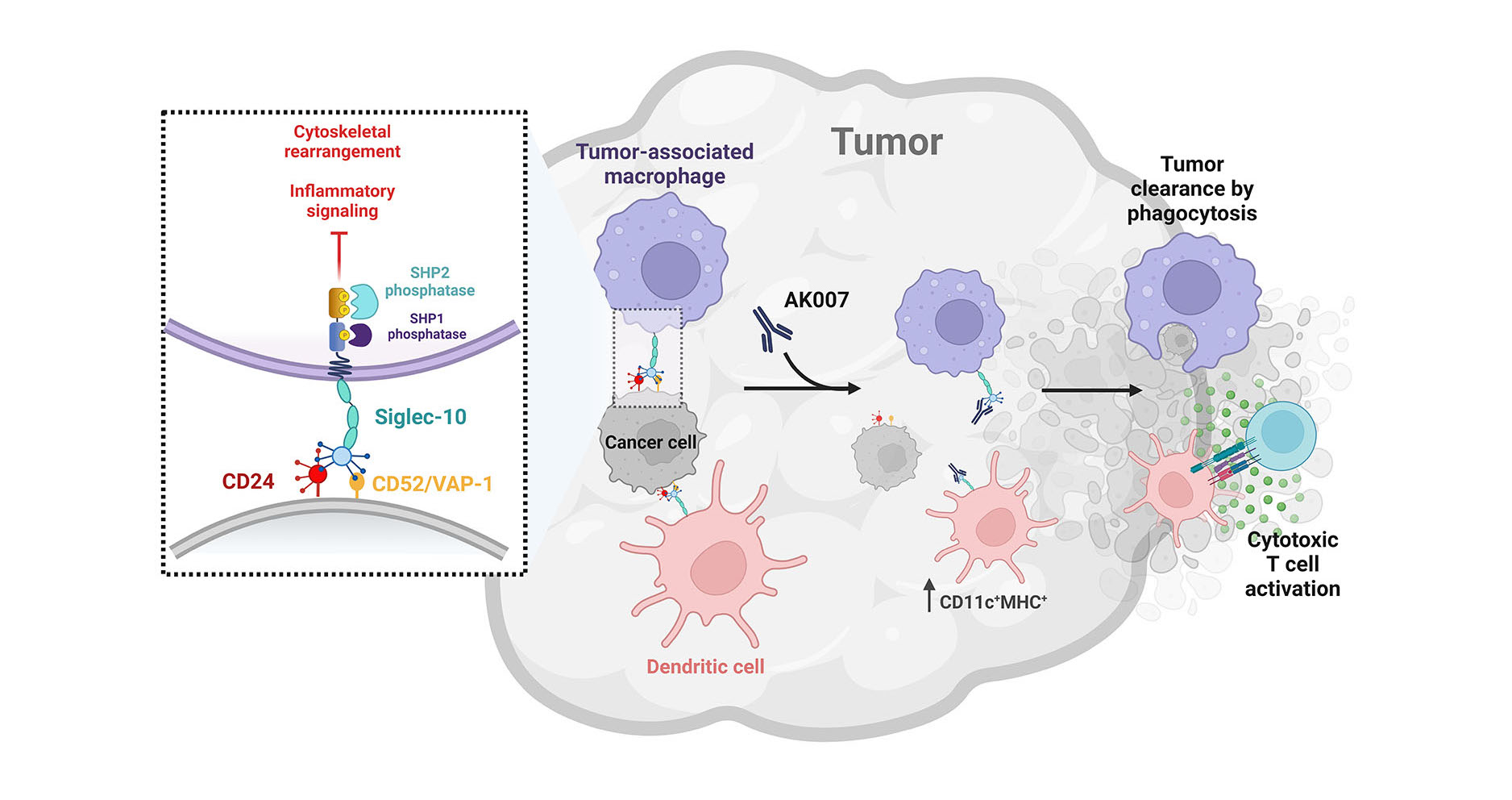
AK007 restores macrophage and dendritic cell anti-tumor activity
AK007 is a humanized Siglec-10 antagonist antibody designed to block Siglec-10 interaction with all known ligands (CD24, CD52, and VAP-1). Preclinical studies have demonstrated that monotherapy treatment with a Siglec-10 antagonist antibody inhibited tumor growth in a syngeneic colon adenocarcinoma model in Siglec-10 transgenic mice. Importantly, this reduced tumor growth was associated with an expansion and activation of TAMs, DCs, and T lymphocytes in the tumor. By targeting Siglec-10, AK007 has the potential to reverse myeloid suppression and promote anti-tumor immunity by directly blocking the checkpoint receptor irrespective of individual ligand interaction.
AK007 monotherapy reduced tumor progression
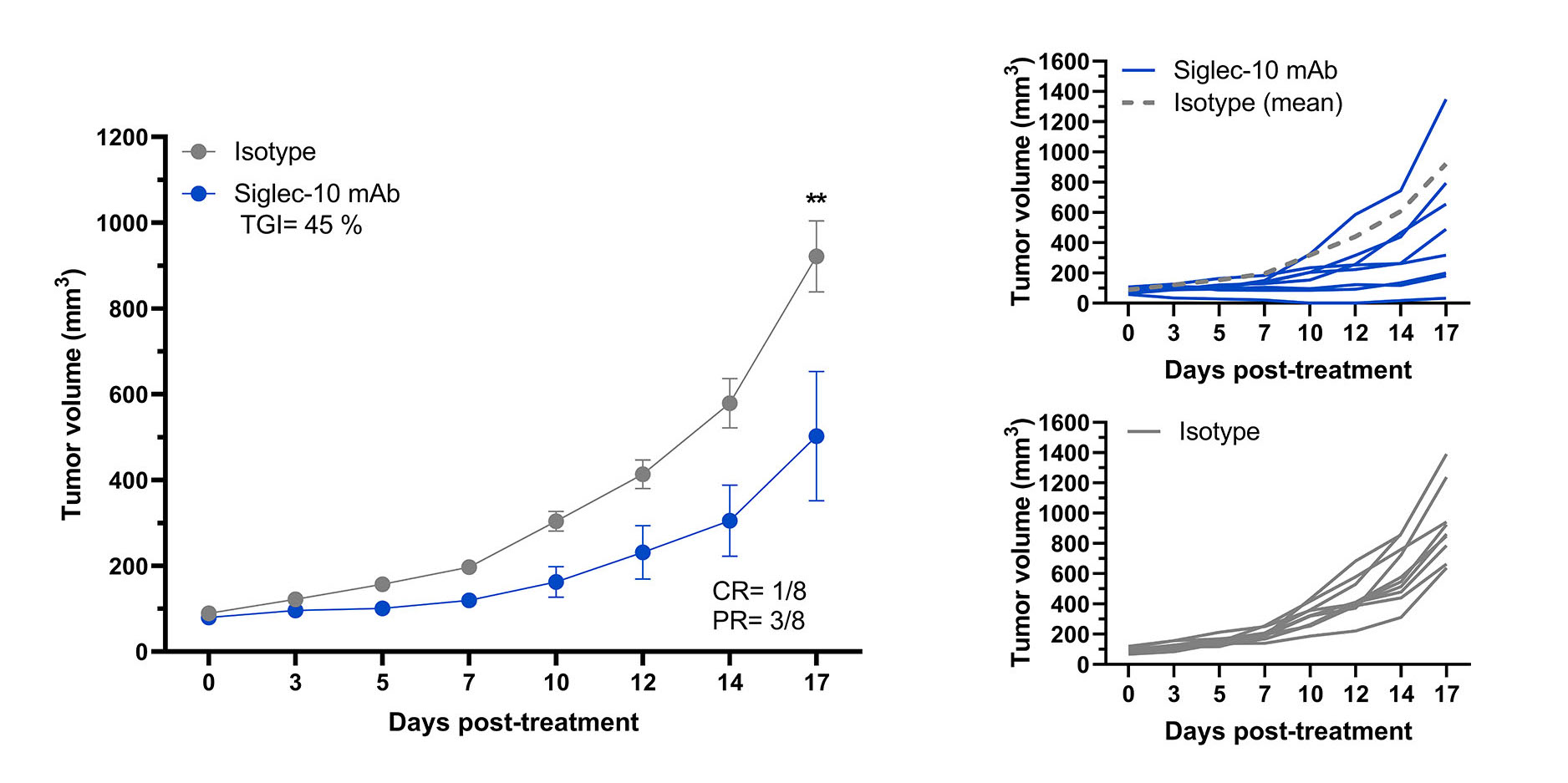
In a Siglec-10 transgenic mouse model, mice were challenged with MC38 tumor cells and tumor volume was monitored twice weekly. Once tumor was established, mice were treated with Siglec-10 mAb (blue), or isotype control (gray) every 2 days for a total of 8 doses. Mice treated with Siglec-10 mAb experienced reduced tumor progression as compared to mice treated with isotype control.
Publications & Presentations
| Date | Focus | Title of Publication |
|---|---|---|
| February 2024 | Preclinical |
American Academy of Allergy Asthma & Immunology (AAAAI) Annual Meeting |
| January 2024 | Preclinical | Regulation of Mast Cells by Overlapping but Distinct Protein Interactions of Siglec-6 and Siglec-8
Allergy |
| October 2023 | Clinical |
Clinical and Translational Gastroenterology |